Device for targeted mini-invasive dermatologic surgery which allows to treat skin disorders and imperfections. The Radio Skin treatment can be safely performed in an outpatient clinic because it is designed to minimize the stress to the tissues and pain during the procedure
![]()
MINI-INVASIVE DERMATOLOGIC SURGERY
Non-surgical blepharoplasty, cherry angiomas, small keloids, seborrheic keratosis, hyperkeratosis, hypertrophic scarring in general, sequelae from acne, skin discolorations, spots, skin fibromas, solar lentiginous, warts, xanthelasma, micro lifting, hyperpigmentation, stretch marks, benign neoplasms of the oral cavity, ear remodeling, etc.
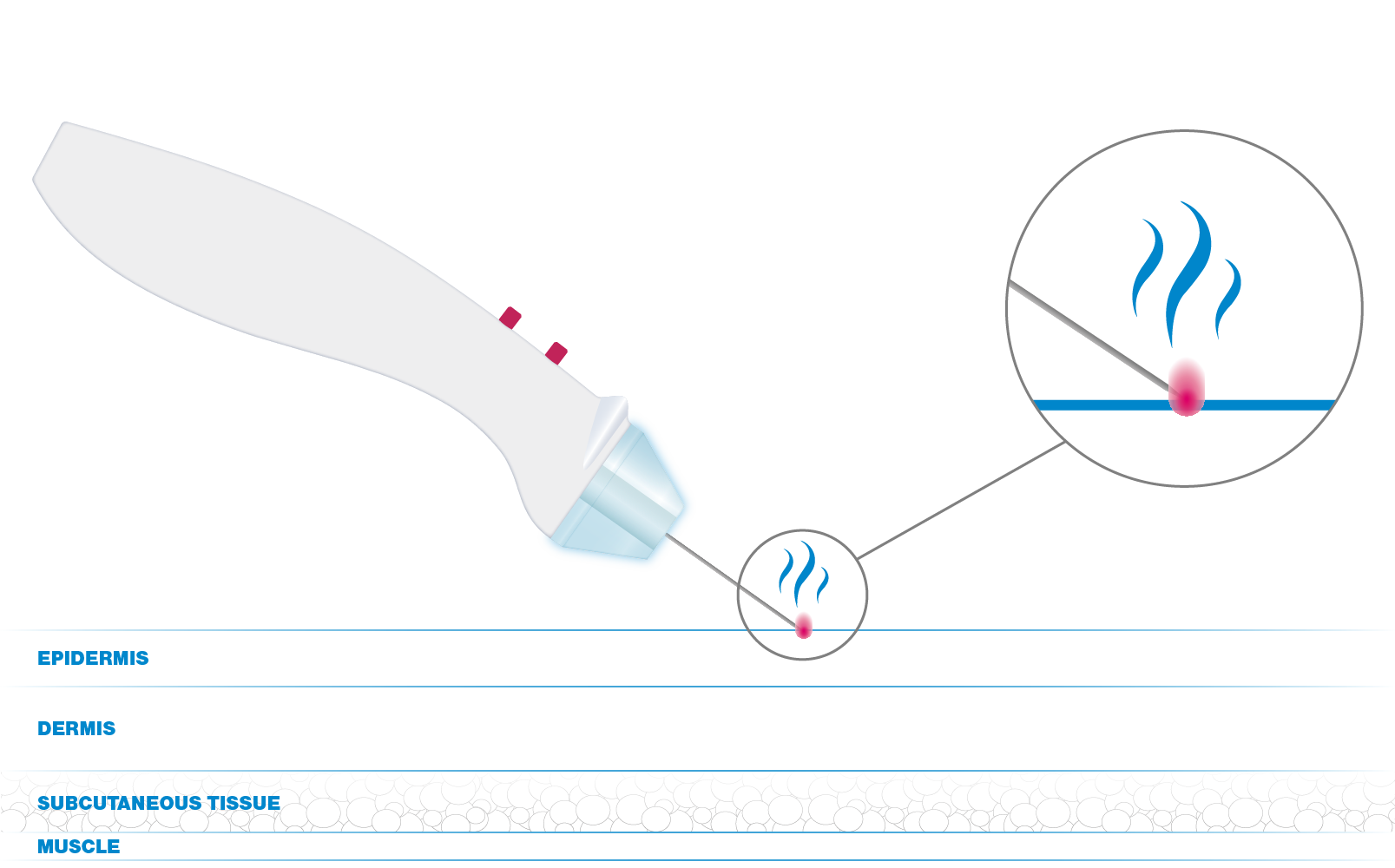
Convoy Electron Flow technology is a particular RF impulse able to create an electric arc between the stratum corneum (tissue with high impedance) and the electrode (the needle).
Said technology allows to convey the energy to the target tissues in order to perform the treatment without having any contact between the skin and the electrode.
The software regulates and stabilises the energy flow, thus resulting in targeted treatments, reduction of recovery time, and functionality without reference electrode.
![]() All of the parameters are displayed on a 15.6” colour touch screen.
All of the parameters are displayed on a 15.6” colour touch screen.
![]()
REMOTE CONNECTIVITY: software update and technical data analysis

